Methods
Would you like to know how neutron scattering can contribute for your company's development and innovation? Contact: industry@sine2020.eu.
For examples of industrial materials investigated with neutrons, please go to our Case Studies page .
Why use neutrons?
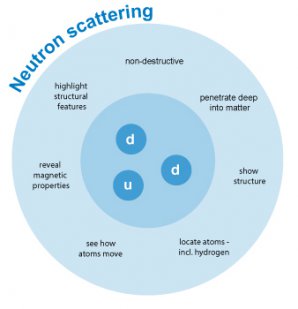
What neutron scattering shows. © R. Bucher, JCNS at MLZ
When beams of neutrons are used to probe small samples of materials they have the power to reveal what cannot be seen using other types of radiation. Neutrons appear to behave either as particles or as waves or as microscopic magnetic dipoles. It is these specific properties which enable them to yield information which is often impossible to obtain using other techniques.
Electrically Neutral
- Neutrons are non-destructive and can penetrate deep into matter. This makes them an ideal probe for biological materials and samples under extreme conditions of pressure, temperature, magnetic field or within chemical reaction vessels.
Microscopically Magnetic
- They possess a magnetic dipole moment which makes them sensitive to magnetic fields generated by unpaired electrons in materials. Precise information on the magnetic behaviour of materials at atomic level can be collected. In addition, the scattering power of a neutron off an atomic nucleus depends on the orientation of the neutron and the spin of the atomic nuclei in a sample. This makes the neutron a powerful instrument for detecting the nuclear spin order.
Ångstrom wavelengths
- Neutron wavelengths range from 0.1 Å to 1000 Å, making them an ideal probe of atomic and molecular structures, be they single atomic species or complex biopolymers.
Techniques
Available industrially relevant techniques are:
- Diffraction : Crystallography and Residual Stress measurements
- Radiography/Imaging/Tomography
- Small angle scattering : (SANS) Characterization of particles and structures in the 1-100 nanometer range
- Spin-echo Small angle scattering (SESANS): Characterization of particles and structures in the 0.1-20 micrometer range
- Reflectometry : Characterization of thin layers in the nanometer range
- Neutron activation analysis (NAA): Non-destructive trace element analysis
- Inelastic Neutron Scattering : Determination of molecular structure in opaque, non-crystalline materials
Applications
The way neutrons scatter off gases, liquids and solid matter gives us information about the structure of these materials (elastic neutron scattering). The neutron excitation of atoms gives information about the binding energy within matter (inelastic neutron scattering). Their ability to act as 'small elementary magnets' makes neutrons an ideal probe for the determination of structures and dynamics of unknown magnetic matter.
Heavy nuclei can be split with neutrons. This can shed light on a number of still unknown processes in atomic fission. Neutrons can also be captured by nuclei. The process releases secondary radiation which can be used to determine the inner structure of these nuclei.
We indicate below how neutrons are used in different fields of research.
Condensed-matter physics, materials science and chemistry
- Examination of the structure of new materials, for example new ceramic high-temperature superconductors or magnetic materials (important for electronic and electrical applications).
- Clarification of still unknown phenomena in processes such as the recharging of electric batteries.
- Storing of hydrogen in metals, an important feature for the development of renewable energy sources.
- Analysis of important parameters (for example elasticity) in polymers (for example plastic material).
- Colloid research gives new information on such diverse subjects as the extraction of oil, cosmetics, pharmaceuticals, food industry and medicine.
- Investigation of micro- and nanostructures.
Biosciences
Biological materials, naturally rich in hydrogen and other light elements, are ideal samples for analysis with neutrons.
- Cell Membranes
- Proteins
- Virus Investigations
- Photosynthesis in Plants
Engineering sciences
Since neutron diffraction is non-destructive, it is ideal for the analysis of different technical phenomena in materials.
- Visualisation of residual stress in materials
- Texture analysis
- Hardening and corrosion phenomena in concrete
- Inhomogeneity and trace elements in materials





