Germany: Deuterated Polymers for Neutron Techniques

P3HT homopolymer (left side) and P3HT-b-PEO diblock colpolymer (right side). The dark color and the metallic shine is typical for P3HT.
Polymers are very versatile compounds used in many different materials e.g. plastics and synthetic rubbers.
Neutron scattering techniques are an effective tool in the study of the physical and chemical properties of polymers as they allow insight into the internal structure and dynamics of the polymer chains. This knowledge helps towards developing new and improved materials.
Neutrons are particularly good at distinguishing between hydrogen atoms and deuterium atoms– an isotope of hydrogen that is very slightly heavier. A technique called contrast variation reveals extra information about polymers if some of the hydrogens normally present are replaced by deuterium. For example, regions of the polymer chain can be selectively labelled and investigated like the chain ends or the middle part.
Unfortunately, the synthesis of deuterated polymers is not easy. The hydrogens cannot easily be directly exchanged with deuterium so deuteration has to occur during the synthesis process. As polymers are created from monomers, these need to be deuterated first. Although some deuterated monomers are available commercially, many useful ones are not, or are prohibitively expensive. The Chemical Deuteration work package of SINE2020 aims to fill this gap in the market.
Andreas Raba and Jürgen Allgaier, at Forschungszentrum Jülich in Germany, have been devising new synthetic routes and procedures to produce deuterated versions of three interesting monomers:
Deuterated isoprene
Structure:
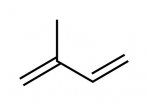
Uses: Synthetic rubber e.g. for tyres
Problem: The established multi-step method for making this monomer is time consuming, provides only a low yield and produces an insufficient degree of deuteration.
Solution: A new synthesis route was developed by adapting an alternative method.
Result: A method to make highly pure isoprene using standard laboratory equipment and suited to producing small-scale amounts. Nearly each different hydrogen position on the molecule can be selectively labelled with deuterium during synthesis by using a deuterated reagent in the respective step of the reaction.
Deuterium labelled polylactic acid (PLA)
Structure:
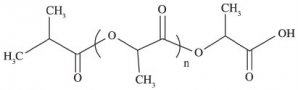
Uses: PLA is a major bio-based plastic on the market. It is biodegradable and suitable for plastics, fibers and biomedical applications. These so-called green polymers from renewable sources could potentially replace petrochemical plastics.
Problem: The standard commercial process of PLA formation process involves three steps. The second step, where lactic acid is converted into lactide, involves high temperatures which hampers deuteration, essential for neutron experiments. It is also important for PLA to be made very pure, have a narrow weight distribution (where the final lengths of each polymer chain are very similar to each other) and also have a high degree of stereocontrol.
Solution: A new method for the conversion of lactic acid-into-lactide step with direct stereoselective conversion using zeolites was found.
Result: The method was first used and adapted on commercial hydrogenous lactic acid. Then it was tried on deuterated lactic acid molecules (produced at ESS for Task 5.4). This worked. In collaboration with Dr Klaus Beckerle at RWTH Aachen the resulting deuterated lactides were then polymerized. The polymer showed narrow weight distribution which indicated a high purity of the synthesized lactides made in this second step.
Deuterium labelled P3HT- b -PEO
Structure:
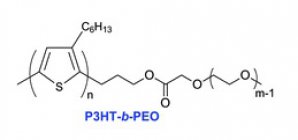
Poly(3-hexylthiophene – b – poly(ethylene oxide)) (P3HT- b -PEO) is a polymer made of two different building blocks called: P3HT and PEO.
Uses: This polymer can conduct electricity and is therefore an interesting material to use in Organic Light Emitting Diodes (OLEDs), organic solar cells, low-cost integrated circuits and organic field-effect transistors (OFETs).
Problem: Known synthetic routes are not optimized to produce a narrow weight distribution of the final polymer which is important for neutron scattering experiments.
Solution: As P3HT- b -PEO is made from the co-polymers P3HT and PEO, the syntheses of these separate molecules and the method of joining them together were optimized.
Result: A synthesis route of P3HT that produced a narrow weight distribution was successfully implemented. Then a ‘click-chemistry’ method to join the co-polymers together was identified as the best route. In order to produce deuterium labelled P3HT – b –PEO, deuteration of the PEO copolymer, using commercially available deuterated ethylene oxide, before joining it to P3HT was the easiest and most economically advantageous solution.
Acknowledgements: Jürgen Allgaier, FZJ
Back to The Road to the ESS
Other articles: Deuteration of biomolecules, Chemical Deuteration, Forschungszentrum Jülich, Deuterated surfactants




